How Breathalyzers Work
We hear and read about drivers who are charged with drunken driving after an accident, and usually a news report on the accident will say what the driver's blood alcohol level was and what the legal limit for blood alcohol is. A driver might be found to have a level of .15, for example, and the legal limit is .08. But what do those figures mean and how do police officers find out if a driver they suspect has been drinking is legally drunk? You have probably heard about the Breathalyzer, but may wonder exactly how a person's breath can show how much that person has had to drink.
It is important for public safety that drunken drivers be taken off the roads. Of the 42,000 traffic deaths in the United States in 1999, about 38 percent were related to alcohol. Drivers who can pass roadside sobriety tests -- they can touch their noses or walk a straight line -- still might be breaking the legal limit for blood alcohol and be a hazard on the road. So police officers use some of the latest technology to detect alcohol levels in suspected drunken drivers and remove them from the streets. Many officers in the field rely on breath alcohol testing devices (Breathalyzer is one type) to determine the blood alcohol concentration, referred to as BAC, in drunken-driving suspects. In this article we examine the scientific principles and technology behind these breath alcohol testing devices.
Need for Testing
Alcohol intoxication is legally defined by the blood alcohol concentration level. However, taking a blood sample in the field for later analysis in the laboratory was not practical or efficient for detaining drivers suspected of driving while impaired (DWI) or driving under the influence (DUI). What was needed was a way to measure something related to BAC without invading a suspect's body. Urine tests for alcohol proved to be just as impractical in the field as blood sampling. In the 1940s breath alcohol testing devices were first developed for use by police. In 1954, Dr. Robert Borkenstein of the Indiana State Police invented the Breathalyzer, one type of breath alcohol testing device used by law enforcement agencies today.
Principle of Testing
Alcohol that a person drinks shows up in the breath because it gets absorbed from the mouth, throat, stomach and intestines into the bloodstream. Alcohol is not digested upon absorption or chemically changed in the bloodstream. As the blood goes through the lungs, some of the alcohol moves across the membranes of the lung's air sacs (alveoli) into the air because alcohol will evaporate from a solution -- that is, it is volatile. The concentration of the alcohol in the alveolar air is related to the concentration of the alcohol in the blood. As the alcohol in the alveolar air is exhaled, it can be detected by the breath alcohol testing device. So instead of having to draw a driver's blood to test his alcohol level, an officer can test the breath on the spot and determine whether there is a reason to arrest the driver.
Because the alcohol concentration in the breath is related to that in the blood, you can figure the BAC by measuring alcohol on the breath. The ratio of breath to blood alcohol is 2,100 to 1. This means that 2,100 milliliters of alveolar air will contain the same amount of alcohol as 1 milliliter of blood.
The legal standard for drunkenness across the United States was .10 for years, but recently many states have adopted the .08 standard. The federal government has pushed states to lower the legal limit. The American Medical Association says that a person can become impaired when the blood alcohol level hits .05. The standard .08 means that there is .08 gram of alcohol per 100 milliliters of blood.
Types of Devices
There are three major types of breath alcohol testing devices based upon different principles:
- A Breathalyzer uses a chemical reaction involving alcohol that produces a color change.
- An Intoxilyzer detects alcohol by infrared (IR) spectroscopy.
- The Alcosensor III or IV detects a chemical reaction of alcohol in a fuel cell.
Regardless of the type, each device has a mouthpiece or tube through which the suspect can blow air and a sample chamber where the air goes. The rest of the device will vary with the type.
Breathalyzer
The device contains a system to sample the breath of the suspect, two glass vials containing the chemical reaction mixture and a system of photocells connected to a meter to measure the color change associated with the chemical reaction. To measure alcohol, a suspect breathes into the device. The breath sample is bubbled in one vial through a mixture of sulfuric acid, potassium dichromate, silver nitrate and water. The principle of the measurement is based upon the following chemical reaction:
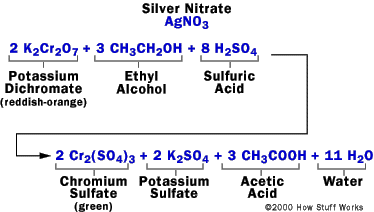
In this reaction,
1. The sulfuric acid removes the alcohol from the air into a liquid solution.
2. The alcohol reacts with potassium dichromate to produce:
- chromium sulfate
- potassium sulfate
- acetic acid
- water
The sulfuric acid also might provide the acidic condition needed for this reaction.
During this reaction, the reddish-orange dichromate ion changes color to the green chromium ion when it reacts with the alcohol; the degree of the color change is directly related to the level of alcohol in the expired air. To determine the amount of alcohol in the expired air, the reacted mixture is compared to a vial of unreacted mixture in the photocell system, which produces an electric current that causes the needle in the meter to move from its resting place. The operator then rotates a knob to bring the needle back to the resting place and reads the level of alcohol from the knob; the more that the operator must turn the knob to return it to rest, the greater the level of alcohol.
Intoxilyzer
This device uses infrared (IR) spectroscopy, which identifies molecules based on the way they absorb IR light. Molecules are constantly vibrating and these vibrations change when they absorb IR light. The changes in vibration include bending and stretching of various bonds. Each type of bond within a molecule absorbs IR at different wavelengths. So, to identify ethanol in a sample, you would have to look at the wavelengths of the bonds in ethanol (C-O, O-H, C-H, C-C) and measure the absorption of IR light. The wavelength helps to identify it as ethanol and the amount of IR absorption tells you how much ethanol is there.
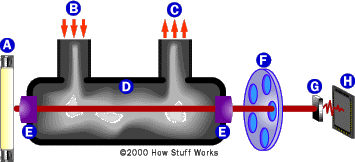
In the Intoxilyzer, a lamp generates a broadband (many wavelengths) IR beam. The IR beam passes through the sample chamber and is focused by a lens onto a spinning filter wheel. The filter wheel contains narrow band filters specific for the wavelengths of the bonds in ethanol. The light passing through each filter is detected by the photocell, where it is converted to an electrical pulse. The electrical pulse is relayed to the microprocessor, which interprets the pulses and calculates the BAC, based on the absorption of infrared light.
Fuel Cell Detectors
Modern fuel cell technology has been applied to breath alcohol detectors. The fuel cell has two platinum electrodes with a porous acid electrolyte material sandwiched between them. As the exhaled air from the suspect flows past one side of the fuel cell, the platinum oxidizes any alcohol in the air to produce acetic acid, protons and electrons. The electrons flow through a wire from the platinum electrode. The wire is connected to an electrical current meter and to the platinum electrode on the other side. The protons move through the lower portion of the fuel cell and combine with oxygen and the electrons on the other side to form water. The more alcohol that becomes oxidized, the greater the electrical current. A microprocessor measures the electrical current and calculates the BAC.
Operators of any breath alcohol testing device must be trained in the use and calibration of the devices, especially if the results are to be used as evidence in DWI trials. Law enforcement officers can carry portable breath testing devices that use the same principle as full-size devices. Court cases can turn on the perceived accuracy of a breath test, however, so prosecutors depend on the results from full-size devices.




